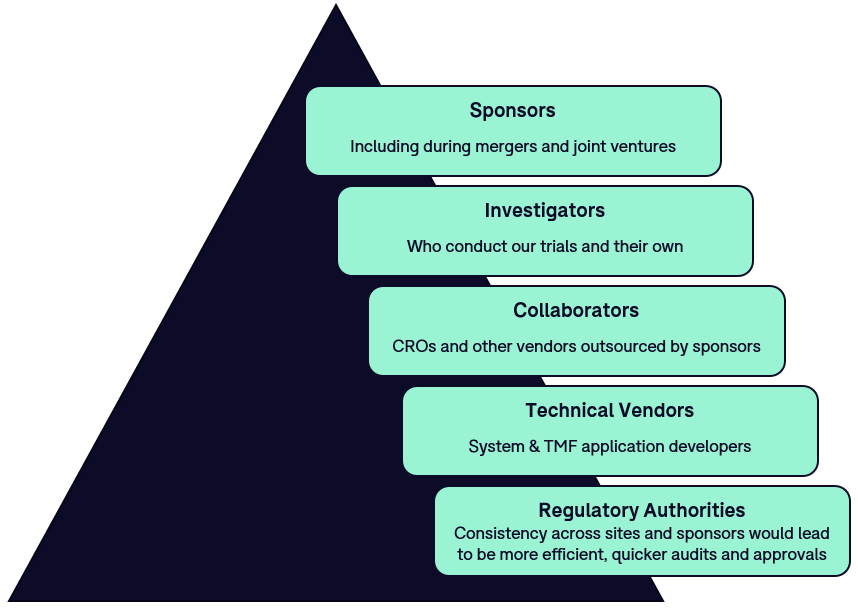
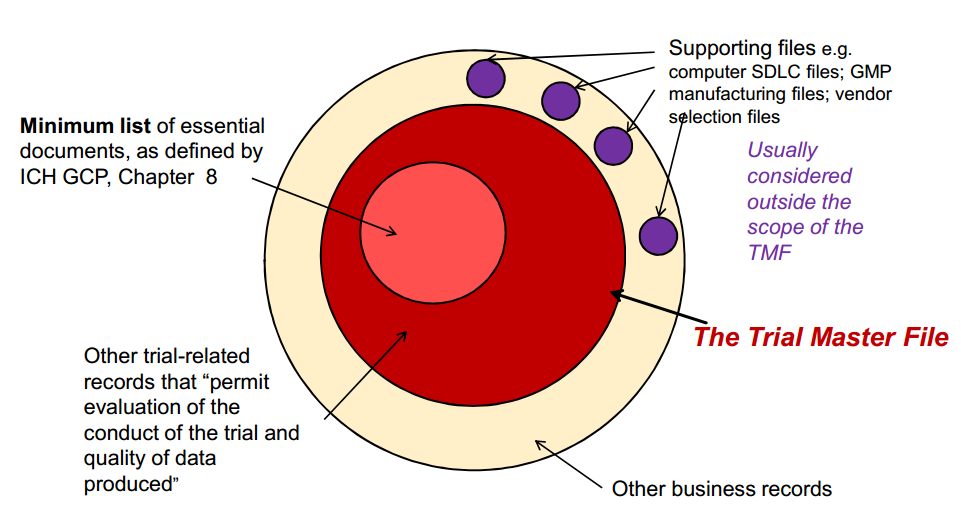
Insights and Action: Takeaways from the DIA Diversity, Equity and Inclusion in the Drug Development Lifecycle Meeting - greenphire.com

DIA Conference: Researchers Work to Balance Data Speed and Accuracy in Complex Studies | 2020-06-21 | CenterWatch

EU-PEARL takes the floor in DIA EUROPE 2022 to share advances made in innovative clinical trials design – EU-PEARL

DrugInfoAssn - The DIA Clinical Trial Conferences are almost around the corner, and one particularity is its JOINT DAY, with both communities joining in these discussions: 🔸 Overview of the EU CTIS (
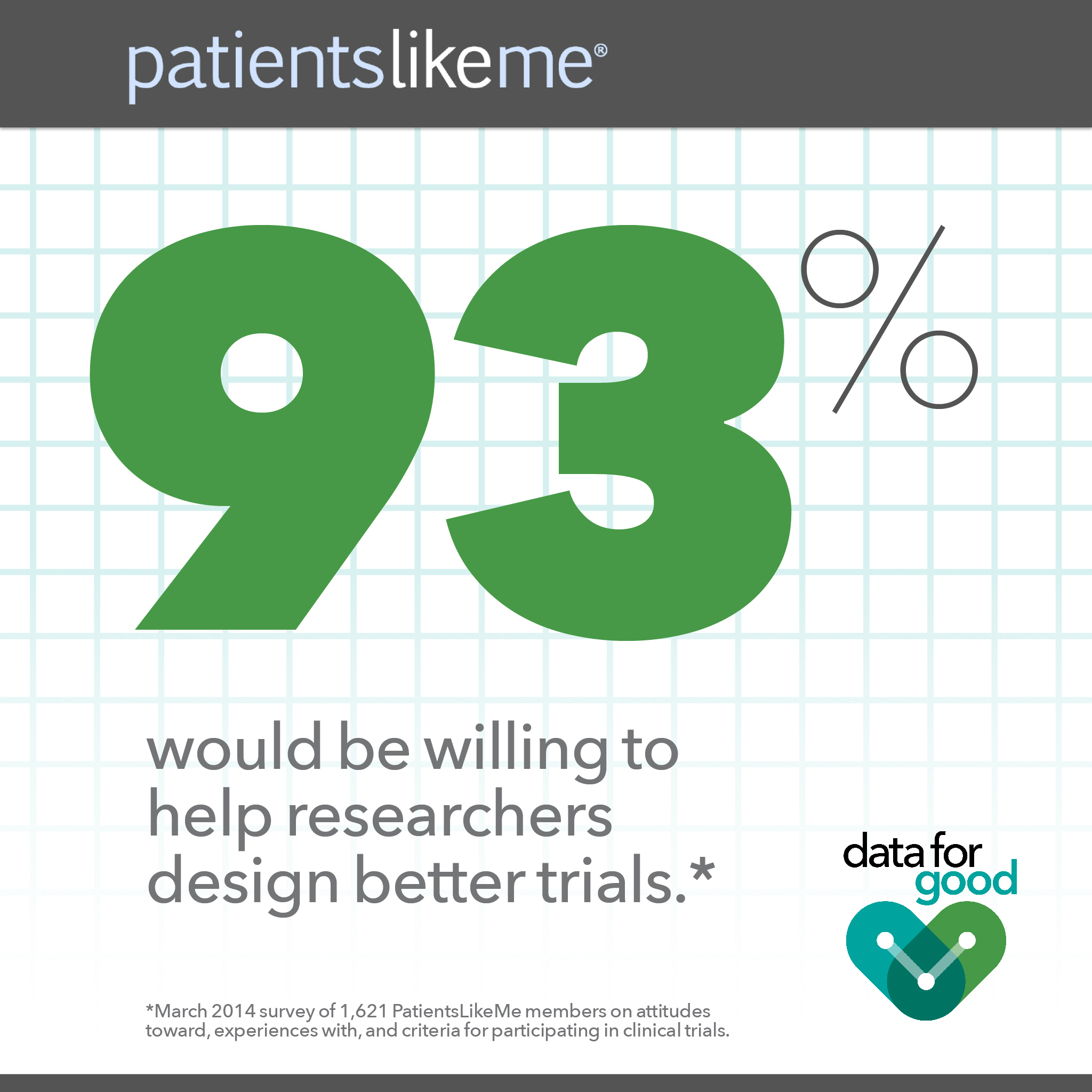
PatientsLikeMe Launches New Services That Make Patients Partners in Medical Research | Business Wire

Staburo @ the DIA Global Clinical Trial Disclosure & Data Transparency Conference 2020 | Data science, biostatistics, data transparency, bioinformatics and statistical programming