IMPD. The Investigational Medicinal Product Dossier IMPD S+P Part Do s and Don't s - PDF Free Download

IMPD review process To ensure the implementation of GCP in the MS of EU... | Download Scientific Diagram

Regulatory Affairs 101: Introduction to Investigational New Drug Applications and Clinical Trial Applications - Chiodin - 2019 - Clinical and Translational Science - Wiley Online Library

Clinical translation of theranostic radiopharmaceuticals: Current regulatory status and recent examples - Kolenc Peitl - 2019 - Journal of Labelled Compounds and Radiopharmaceuticals - Wiley Online Library

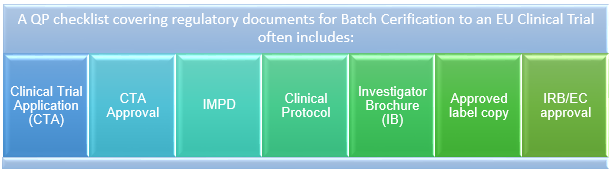
What Are the Documents Required for Clinical Trial Applications to Regulatory Authorities in Europe? - Sofpromed

IMPD. The Investigational Medicinal Product Dossier IMPD S+P Part Do s and Don't s - PDF Free Download

About OMICS Group OMICS Group is an amalgamation of Open Access Publications and worldwide international science conferences and events. Established in. - ppt download

What Are the Documents Required for Clinical Trial Applications to Regulatory Authorities in Europe? - Sofpromed

IMPD review process To ensure the implementation of GCP in the MS of EU... | Download Scientific Diagram

Pre Clinical Assessment - Woodley BioReg Regulatory Affairs, Compliance and Conformance for pharmaceutical, biopharmaceutical, healthcare, API and Medical device manufacturers

What you Need and When – The Key Documents in the Drug Lifecycle - Trilogy Writing & Consulting GmbH