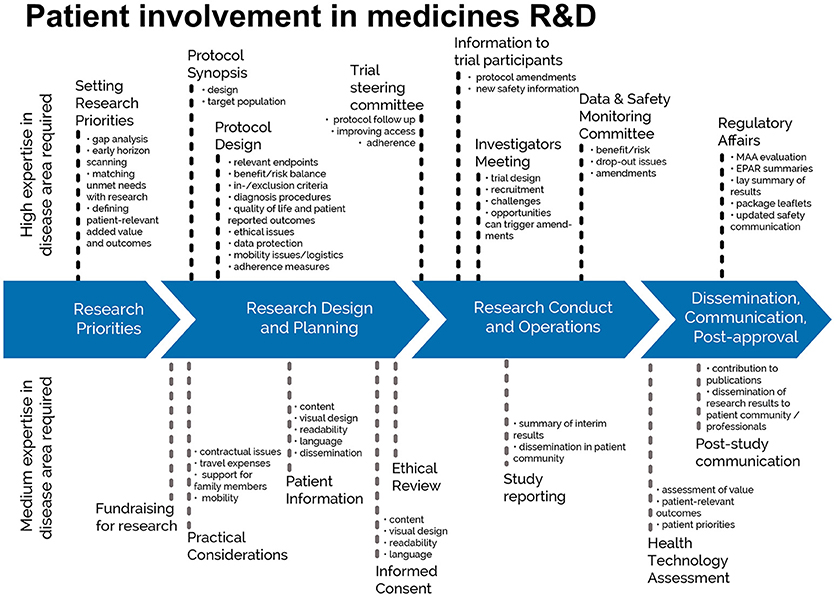
Cromos Pharma - Are you a sponsor seeking to conduct clinical trials in the European Union but do not have a registered office in the European Economic Area? If so, in order
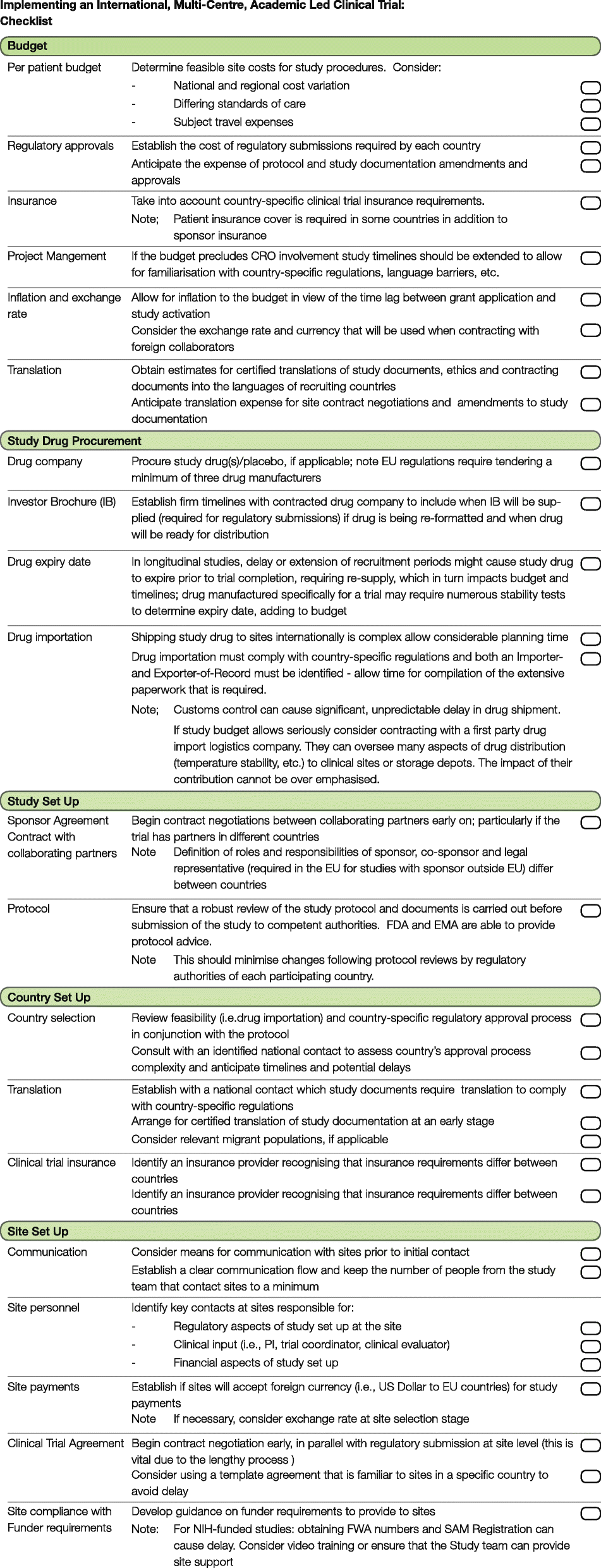
A checklist for clinical trials in rare disease: obstacles and anticipatory actions—lessons learned from the FOR-DMD trial | Trials | Full Text

What are the requirements for the legal representative of a non EU-sponsor in view of article 74 of – Clinical Consulting