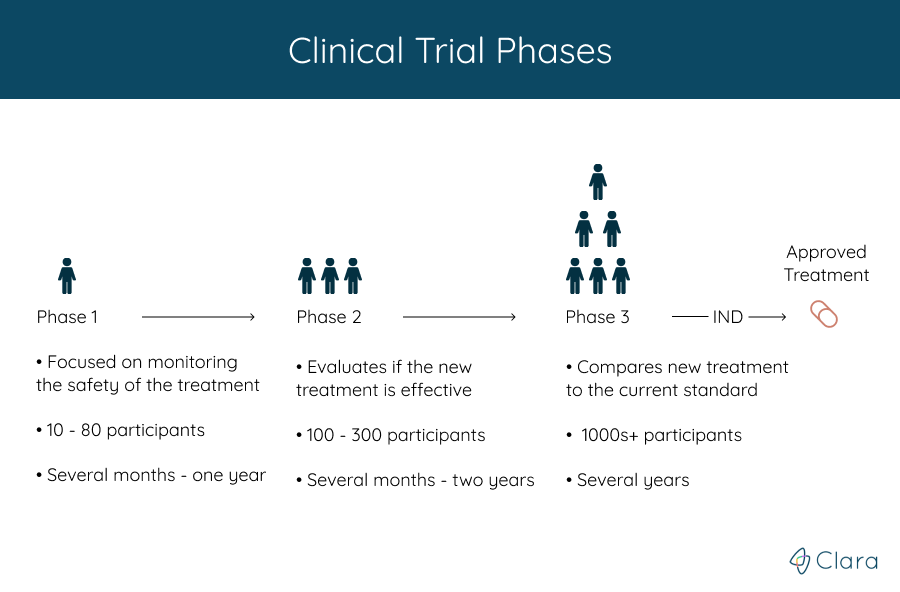
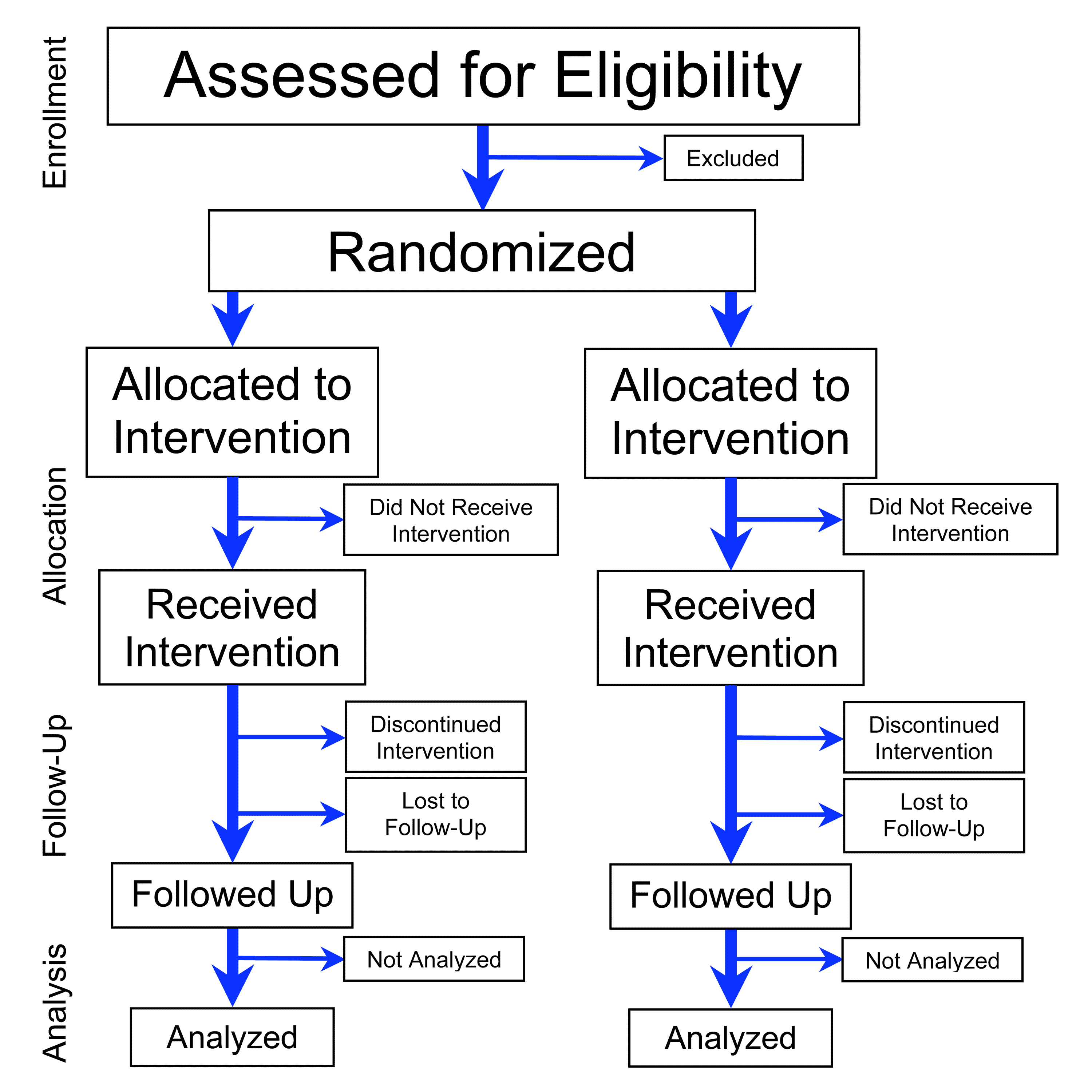
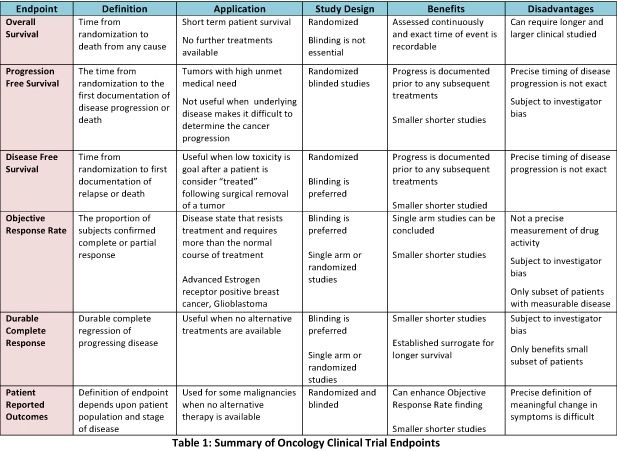
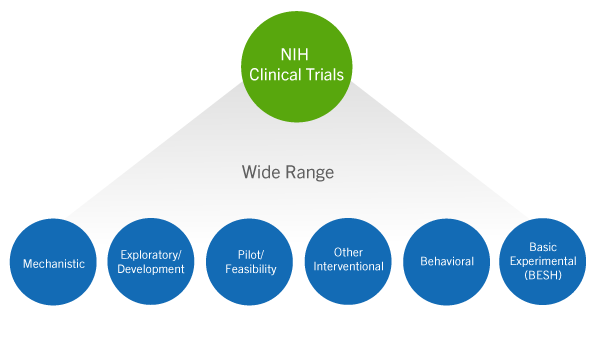
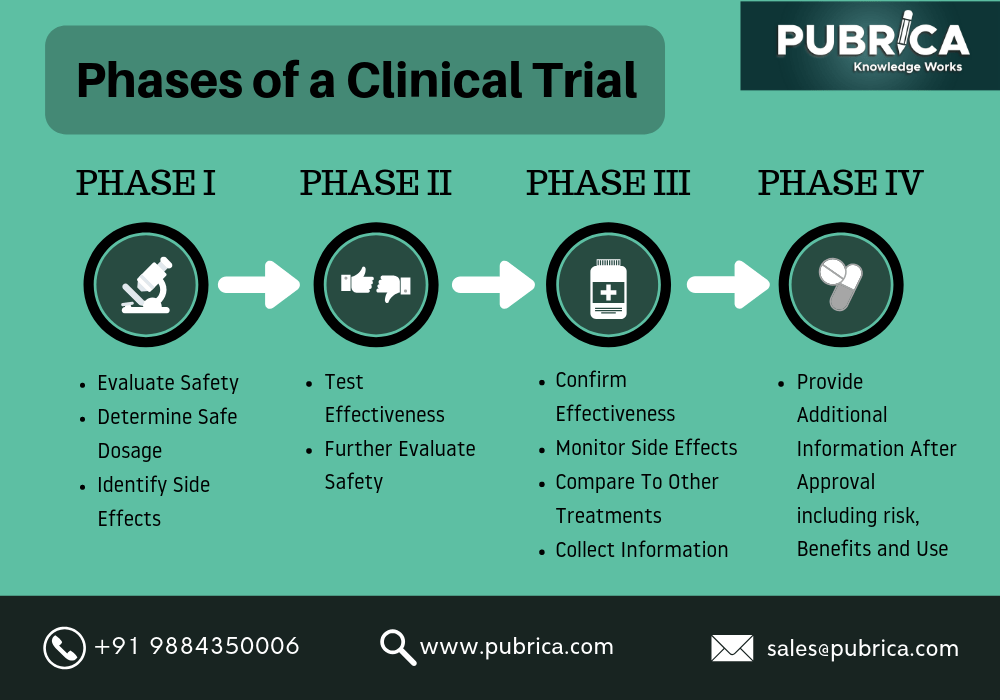
Understanding Clinical Trial Terminology: What's a Phase 1, 2 or 3 Clinical Trial? - Concert Pharmaceuticals

Regulatory Clinical Trials Clinical Trials. Clinical Trials Definition: research studies to find ways to improve health Definition: research studies to. - ppt download

Safety and Immunogenicity of a DNA SARS-CoV-2 vaccine (ZyCoV-D): Results of an open-label, non-randomized phase I part of phase I/II clinical study by intradermal route in healthy subjects in India - eClinicalMedicine

PHASE II STUDY DESIGNS: PART 2 Resident and Fellows Lecture Series April 12, 2016 Elizabeth Garrett-Mayer, PhD Hollings Cancer Center. - ppt download